UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO SECTION 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the Month of January 2022
Commission File Number: 001-41157
BIONOMICS LIMITED
(Exact Name of Registrant as Specified in Its Charter)
200 Greenhill Road
Eastwood SA 5063
Australia
Tel: +618 8150 7400
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
INFORMATION CONTAINED IN THIS REPORT ON FORM 6-K
On January 9, 2022, Bionomics Limited (the “Company”) lodged a presentation with the Australian Securities Exchange (the “ASX”), as required by the laws and regulations of Australia, that it presented at the H.C. Wainwright BioConnect Conference. The presentation is furnished herewith as Exhibit 99.1 to this report on Form 6-K.
The information set forth in the paragraph above shall not be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, or under the Securities Exchange Act of 1934, whether made before or after the date hereof, except as expressly provided by specific reference in such a filing.
The furnishing of the attached presentation is not an admission as to the materiality of any information therein. The information contained in the corporate presentation is summary information that is intended to be considered in the context of more complete information included in the Company’s filings with the Securities and Exchange Commission (the “SEC”) and other public announcements that the Company has made and may make, by press release or otherwise, from time to time. The Company undertakes no duty or obligation to update or revise the information contained in this report, although it may do so from time to time as its management believes is appropriate. Any such updating may be made through the filing or furnishing of other reports or documents with the SEC, through press releases, by updating its website or through other public disclosures.
Exhibits
| 99.1 | Bionomics Presentation for H.C. Wainwright BioConnect 2022 |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Bionomics Limited | ||||||
| Date: January 12, 2022 | By: | /s/ Errol De Souza | ||||
| Name; | Errol De Souza, Ph.D. | |||||
| Title: | Executive Chairman | |||||

Exhibit 99.1 TO IMPROVE THE LIVES OF PATIENTS WITH SERIOUS CNS DISORDERS Corporate Presentation Nasdaq: BNOX ASX: BNO H.C. Wainwright BIOCONNECT Virtual Conference January 10 - 13, 2022

SAFE HARBOR STATEMENT 2 Factors Affecting Future Performance This presentation may contain forward-looking statements within the meaning of the United States’ Private Securities Litigation Reform Act of 1995. Any statements contained in this presentation that relate to prospective events or developments, including, without limitation, statements made regarding Bionomics’ drug candidates (including BNC210, BNC105, BNC101 and BNC375), its licensing agreement with Merck & Co. and any milestone or royalty payments thereunder, drug discovery programs, ongoing and future clinical trials, and timing of the receipt of clinical data for our drug candidates are deemed to be forward-looking statements. Words such as believes, anticipates, plans, expects, projects, forecasts, will and similar expressions are intended to identify forward-looking statements. There are a number of important factors that could cause actual results or events to differ materially from those indicated by these forward-looking statements, including unexpected safety or efficacy data, unexpected side effects observed in clinical trials, risks related to our available funds or existing arrangements, delays or difficulties associated with conducting clinical trials, our failure to introduce new drug candidates or platform technologies or obtain regulatory approvals in a timely manner or at all, regulatory changes, inability to protect our intellectual property, risks related to our international operations, as well as other factors. Results of studies performed on our drug candidates and competitors’ drugs and drug candidates may vary from those reported when tested in different settings. The inclusion of forward-looking statements should not be regarded as a representation by Bionomics that any of its expectations, projections or plans will be achieved. Actual results may differ from those expectations, projections or plans due to the risks and uncertainties inherent in Bionomics business and other risks described in Bionomics’ filings with the SEC. New risk factors emerge from time to time and it is not possible for our management to predict all risk factors, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in, or implied by, any forward-looking statements. You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this presentation. Subject to the requirements of any applicable legislation or the listing rules of any stock exchange on which our securities are quoted, we disclaim any intention or obligation to update any forward-looking statements as a result of developments occurring after the date of this presentation. Certain information contained in this presentation relates to, or is based on, studies, publications, surveys and other data obtained from third party sources and Bionomics’ own internal estimates and research. While we believe these third party sources to be reliable as of the date of this presentation, we have not independently verified, and make no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while we believe our own internal research is reliable, such research has not been verified by any independent source.

Emerging Leader in Neuropsychiatry 3 Diversified, clinical-stage biopharmaceutical company developing novel, allosteric ion channel modulators designed to transform the lives of patients suffering from serious Central Nervous System (CNS) disorders 1 BNC210 in ongoing Phase 2 for acute treatment in Social Anxiety Disorder (SAD) – Established clinical PoC in GAD and Fast Track designation from FDA for SAD BNC210 in ongoing Phase 2b ATTUNE trial with Fast Track designation from FDA for PTSD Large underserved markets with over 22 million patients in the United States alone suffering from SAD and PTSD and no new FDA approved therapies in nearly two decades Strategic partnership with Merck & Co. for cognitive impairment in Alzheimer’s disease and other CNS conditions Pipeline of partnering candidates targeting potassium (Kv) and sodium (Nav) ion channels Well-capitalized balance sheet with multiple potential near term value-driving milestones PoC = Proof of Concept GAD = Generalized Anxiety Disorder PTSD = Post-Traumatic Stress Disorder 1. Wise et al 2020, Biological Psychiatry; Perkins et al 2021, Molecular Psychiatry
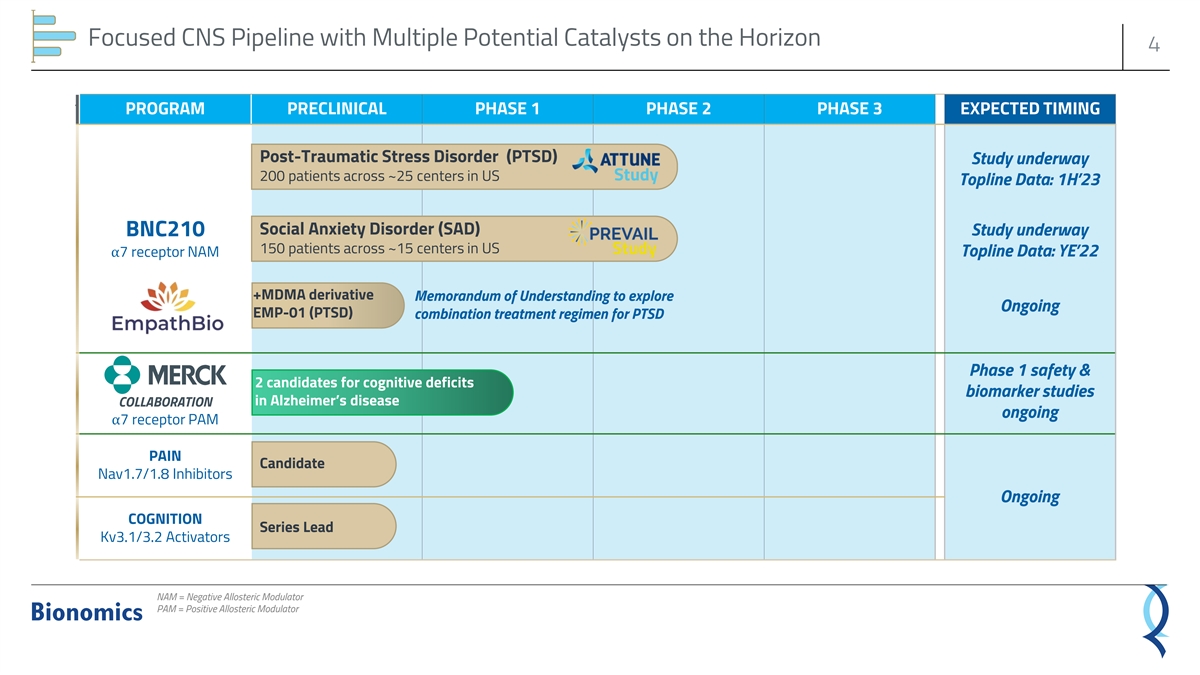
Focused CNS Pipeline with Multiple Potential Catalysts on the Horizon 4 ` PROGRAM PRECLINICAL PHASE 1 PHASE 2 PHASE 3 EXPECTED TIMING Post-Traumatic Stress Disorder (PTSD) Study underway 200 patients across ~25 centers in US Study Topline Data: 1H’23 Social Anxiety Disorder (SAD) BNC210 Study underway 150 patients across ~15 centers in US Study Topline Data: YE’22 α7 receptor NAM +MDMA derivative Memorandum of Understanding to explore Ongoing EMP-01 (PTSD) combination treatment regimen for PTSD Phase 1 safety & 2 candidates for cognitive deficits biomarker studies in Alzheimer’s disease COLLABORATION ongoing α7 receptor PAM PAIN Candidate Nav1.7/1.8 Inhibitors Ongoing COGNITION Series Lead Kv3.1/3.2 Activators NAM = Negative Allosteric Modulator PAM = Positive Allosteric Modulator

A Differentiated Approach: The a7 Nicotinic Acetylcholine Receptor 5 Normalizing Effect Utilizing Allosteric Modulation Targeting Distinct CNS Conditions with Neurotransmitter Imbalance 1 2 2+ Ca flow Channel opens: through the 2+ Ca ACh binds to channel when BNC 210 (NAMs) a7 receptor a7 receptors + Anxiety orthosteric + PTSD are activated sites + Depression Hypercholinergic Disease States Transmembrane Binding Domain Hypocholinergic Disease States 3 4 PAM PAMs / NAMs Allosteric drug Indications bind to a7 transmembrane binding 2+ receptor Ca domain restores normal allosteric sites activity 2+ Ca = Calcium ions ACh = Acetylcholine NAM = Negative Allosteric Modulator PAM = Positive Allosteric Modulator Cholinergic = System associated with memory, selective attention, and emotional processing cognitive functions PTSD = Post-Traumatic Stress Disorder CIAS = Cognitive Impairment Associated with Schizophrenia ADHD = Attention Deficit Hyperactivity Disorder

Harnessing the Power of a Novel Neuromodulatory Mechanism 6 ANXIETY & NORMAL Action of BNC210 PTSD depends on Acetylcholine neurotransmission NORMAL BNC210 RESTORES and Allosteric ACh MOOD NORMAL MOOD 2+ Ca Modulation of BNC210 MOOD DISORDERS a7 nAChR NAMs have self-limiting activity determined by the cooperative interaction between BNC210 and Acetylcholine binding at the allosteric and orthosteric sites, respectively nAChR = Nicotinic Acetylcholine Receptor NAM = Negative Allosteric Modulator 2+ Ca = Calcium ions ACh = Acetylcholine

7 BNC210 in Social Anxiety Disorder

Targeting a Large Segment of the Anxiety Market 8 Acute Anxiety in SAD Represents a Significant Unmet Need Social Anxiety Disorder (SAD), or Social Phobia, is a significant and persistent fear of social and ~31M performance-related situations 12.1% of adults at some point Includes anxiety from everyday social situations as in their lives well as “Fear of Public Speaking” ~18M 7.1% A disorder that substantially impacts many people’s prevalence daily lives in adults ~7M • Amongst the largest mental health conditions with lifetime 36% of adults >10 years SAD prevalence affecting >31M Americans symptoms • No FDA-approved fast-acting medications for as-needed Unmet medical need to large patient population treatment Advancement in care No FDA-approved fast-acting competition • Medications with the right pharmacokinetic profile and a Ability to potentially achieve large market share novel mechanism are needed Sources: US Census Bureau. https://www.census.gov/library/stories/2021/08/united-states-adult-population-grew-faster-than-nations-total-population-from-2010-to-2020.html NIMH. Social Anxiety Disorder data from 2017 National Comorbidity Survey (NCS). https://www.nimh.nih.gov/health/statistics/social-anxiety-disorder.shtml Anxiety and Depression Association of America (ADAA). Social Anxiety Disorder - Understand the Facts https://adaa.org/understanding-anxiety/social-anxiety-disorder

BNC210 Significantly Reduced Panic Symptoms in Humans: CCK-4-Induced Model 9 Total # of Panic Symptoms Panic Symptom Intensity 100 100 90 90 -37.7% -52.7% Placebo- 80 80 (p<0.048) (p<0.041) 70 70 controlled study 60 60 50 50 in 15 healthy 40 40 30 volunteers who 30 20 20 experienced a 10 10 0 0 CCK-4-induced Placebo BNC210 panic attack BNC210 demonstrated statistically significant reduction in both number and intensity of panic symptoms measured with the Panic Symptom Scale CCK-4 = Cholecystokinin Tetrapeptide (a peptide that induces anxiety and panic symptoms) Panic Symptom Scale (% of Placebo) Panic Symptom Scale (% of Placebo)

BNC210 Phase 2 in GAD Observed to Have Acute Anxiolytic Activity 10 Performed BNC210 300 mg Fearful ‘JORT’ Faces BNC210 2000 mg Lorazepam 1.5 mg 24 GAD Patients Placebo fMRI Monitoring Significantly reduced activation of Significantly reduced connectivity Significantly reduced threat avoidance L & R amygdala caused by viewing between amygdala and ACC while behavior of anxious subjects in the JORT fearful faces (L: p<0.05; R: p<0.01) viewing fearful faces (p<0.05) behavioral task 300 mg 300 mg • Amygdala activation is an imaging surrogate for anxiety • Connectivity between the amygdala and Anterior Cingulate Cortex (ACC) is very strong in high anxiety = BNC210 Wise T. et al., Biological Psychiatry 2020 (https://doi.org/10.1016/j.biopsych.2019.12.013); Perkins A. et al., Translational Psychiatry 2021 (https://doi.org/10.1038/s41398-020-01141-5) GAD = Generalized Anxiety Disorder JORT = Joystick Operated Runway Task fMRI = Functional Magnetic Resonance Imaging 4 WAY CROSSOVER

BNC210 Addresses the Shortcomings of Existing Social Anxiety Disorder Approaches 11 CURRENT TREATMENTS FOR SOCIAL ANXIETY DISORDER Large Market NO NO NO FAST NO WITHDRAWAL MEMORY MOTOR Potential for Social DRUG ACTING SEDATION SYNDROME IMPAIRMENT IMPAIRMENT Anxiety Disorder 1 Benzodiazepines X X X X Attractive Clinical Novel Mechanism 2 BNC210 & Regulatory SSRIs / SNRIs of Action Potential X X Pathway Potential for Acute Treatment in Social Anxiety Disorder Patients BNC210 IS DESIGNED TO PROVIDE ADVANTAGES COMPARED TO CURRENT THERAPIES* Anti-Anxiety Effects NO NO NO Rapid Onset FAST In healthy subjects NO WITHDRAWAL MEMORY MOTOR DRUG ACTING SEDATION SYNDROME IMPAIRMENT IMPAIRMENT (anti-panic) and GAD of Action patients (anti-anxiety) BNC 210 * Potential benefits based on analysis of data from separate studies and not on results that might have been obtained from head-to-head studies. Such data may not be directly comparable due to differences in study protocols, conditions and patient populations. Accordingly, cross-trial comparisons may not be reliable predictors of the relative efficacy or other benefits of BNC210 compared to existing therapies or other product candidates that may be approved or are in development for the treatment of PTSD or SAD. 1. Includes Valium and certain other benzodiazepines 2. Includes Prozac and certain other SSRIs (Selective Serotonin Reuptake Inhibitors) / SNRIs (Serotonin-Norepinephrine Reuptake Inhibitors)

BNC210’s Rapid Onset of Action is Well-Positioned for Social Anxiety Disorder 12 Emerging Regulatory Landscape & Unmet Need Rapid Onset of Action with BNC210 Formulation ✓✓ • Clinically demonstrated potential for reducing anxiety in acute • No fast-acting FDA-approved medications for treatment of GAD patients and following panic induction as-needed treatment of SAD • Observed acute anxiolytic efficacy of BNC210 similar to lorazepam without sedative properties and addiction liability • Benzodiazepines prescribed off-label have significant side effects of sedation, cognitive • Formulation well-suited for acute dosing – Rapidly absorbed to impairment and potential for addiction high concentrations within a short period of time • Growing unmet need based on improving awareness and evolving social dynamics Maximum • FDA precedent on simplified public speaking concentrations challenge endpoint for acute anxiety reduction reached in vs. placebo* ~45 – 105 min. across the dose range *Based on path of CNS peer proceeding with registrational Phase 3 endpoint

BNC210 Phase 2 PREVAIL Social Anxiety Disorder Trial 13 Acute Social Anxiety Disorder Study Highlights Phase 2 PREVAIL Study Design SCREENING LIEBOWITZ SOCIAL ANXIETY SCALE ✓ Potential to conduct a cost-effective trial with LSAS ≥ 70 >95: Very severe social phobia an efficacy endpoint conducive to rapid data 80–95: Severe social phobia generation 65–80: Marked social phobia RANDOMIZATION (DOUBLE-BLIND) 55–65: Moderate social phobia ✓ Ability to leverage development strategies ~50 ~50 ~50 15 – 20 CENTERS 225 mg 675 mg of other Social Anxiety Disorder public CNS BNC 210 BNC 210 PLACEBO trial designs 60min 60min 60min EFFICACY ENDPOINT ✓ Received FDA clearance for IND filing and SUBJECTIVE UNITS OF SINGLE ANXIETY CHALLENGE DISTRESS SCALE (SUDS): FDA Fast Track designation Measures the self-reported ✓ intensity of anxiety and/or û distress in SAD patients ✓ Phase 2 trial underway and will read out topline data by end of 2022 FDA Fast Track designation Topline data expected YE’22 LSAS = Liebowitz Social Anxiety Scale

14 BNC210 in Post-Traumatic Stress Disorder

Tackling the Profound Disease Burden of Post-Traumatic Stress Disorder 15 PTSD Represents a Significant Unmet Need ✓ 70% of people will experience a traumatic event in their lifetime, but most people recover normally ~21M 8% of adults at ✓ PTSD results from exposure to actual or threatened some point in death, serious injury or sexual violence their lives ~9M ✓ PTSD affects up to 8% of adults during their 3.4% 1 lifetime prevalence in adults ✓ PTSD is a global mental health problem that is ~7M associated with significant morbidity and mortality 75% of adults inadequately and shows up in all facets of peoples’ lives 2 treated ✓ No newly approved pharmacotherapy in almost Unmet medical need to large patient population two decades Advancement in care No branded competition ✓ Medications with a novel mechanism of action that Ability to potentially achieve large market share can address the pathophysiology of PTSD are needed 1. Kilpatrick, D., Resnick, H., Milanak, M., Miller, M., Keyes, K. and Friedman, M., 2013. National Estimates of Exposure to Traumatic Events and PTSD Prevalence Using DSM-IV and DSM-5 Criteria. Journal of Traumatic Stress, 26(5), pp.537–547; 2 Mayo LM, Asratain A., Lindé J et al. Elevated Anandamide, Enhanced Recall of Fear Extinction, and Attenuated Stress Responses Following Inhibition of Fatty Acid Amide Hydrolase: A Randomized, Controlled Experimental Medicine Trial. Biol Psychiatry. 2020 Mar 15; 87(6): 538-54 2. Only 20 to 30% of PTSD patients achieve clinical remission on SSRI therapies. US Census Bureau. https://www.census.gov/library/stories/2021/08/united-states-adult-population-grew-faster-than-nations-total-population-from-2010-to-2020.html

BNC210 Anti-Anxiety Signals Observed in Animal Models and Human Translational Studies 16 Conditioned Fear Extinction Model Emotional Visual Analog Scale (eVAS) People with anxiety disorders and PTSD have amplified fear responses to trauma- or stress-related stimuli and impaired fear BNC210 enhanced emotional recovery BNC210 enhanced fear extinction extinction following a CCK-induced panic attack following conditioned response training *Time in minutes after CCK-4 injection CCK-4 = Cholecystokinin Tetrapeptide (a peptide that induces anxiety and panic symptoms) eVAS = Emotional Visual Analog Scale

BNC210 is Potentially Well-Positioned in Post-Traumatic Stress Disorder 17 Study ✓ Anti-depressant and ✓ Pharmacometric ✓ New tablet formulation ✓ Type C meeting with ✓ Phase 2b ATTUNE trial anti-anxiety trends seen analysis of Phase 2 overcomes food effect FDA started in July 2021 at earlier time points PTSD data of suspension ✓ FDA granted Fast Track ✓ Topline data expected formulation ✓ Safety profile generally ✓ Predicted significant designation in PTSD 1H 2023 well tolerated ✓ Achieved exposure efficacy potential with Did not meet primary target predicted from adequate drug exposure endpoint*; lower than pharmacometric achieved expected exposure of analysis liquid suspension formulation✓ Extended IP coverage *Primary endpoint of CAPS-5 total symptom severity score at 12 weeks
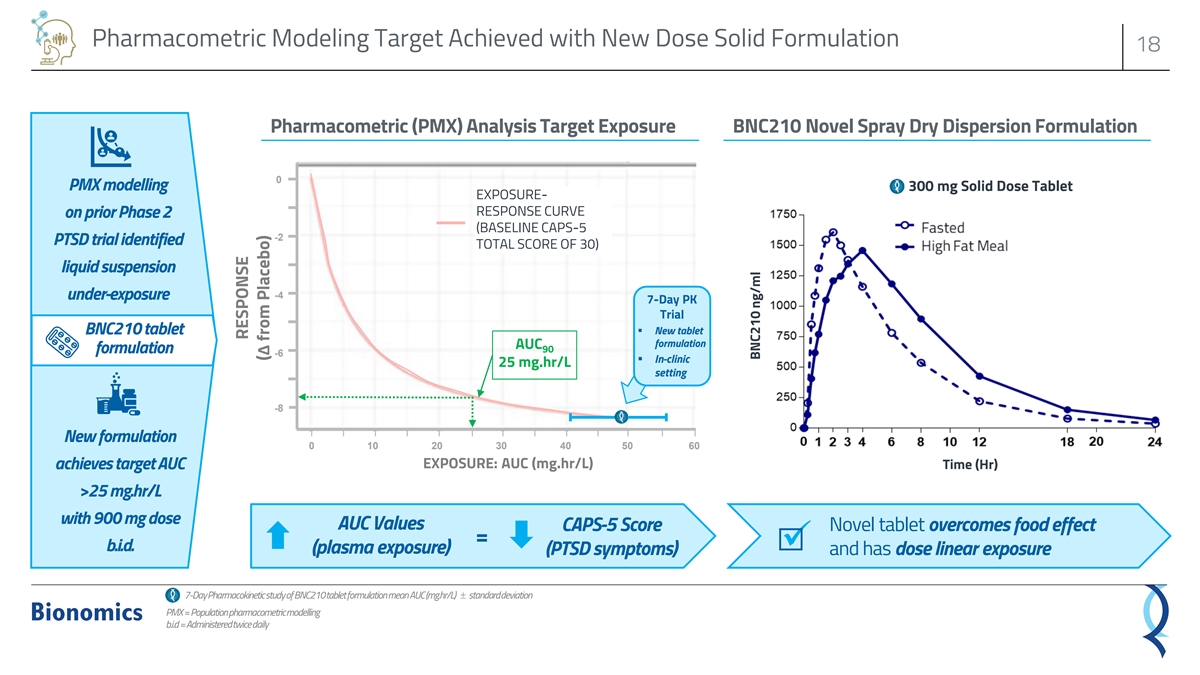
Pharmacometric Modeling Target Achieved with New Dose Solid Formulation 18 Pharmacometric (PMX) Analysis Target Exposure BNC210 Novel Spray Dry Dispersion Formulation 0 PMX modelling 300 mg Solid Dose Tablet EXPOSURE- RESPONSE CURVE on prior Phase 2 (BASELINE CAPS-5 -2 PTSD trial identified TOTAL SCORE OF 30) liquid suspension -4 under-exposure 7-Day PK Trial BNC210 tablet▪ New tablet formulation AUC 90 formulation -6 ▪ In-clinic 25 mg.hr/L setting -8 New formulation 0 10 20 30 40 50 60 EXPOSURE: AUC (mg.hr/L) Time (Hr) achieves target AUC >25 mg.hr/L with 900 mg dose AUC Values CAPS-5 Score Novel tablet overcomes food effect = b.i.d.✓ (plasma exposure) (PTSD symptoms) and has dose linear exposure 7-Day Pharmacokinetic study of BNC210 tablet formulation mean AUC (mg.hr/L) ± standard deviation PMX = Population pharmacometric modelling b.i.d= Administered twice daily RESPONSE (Δ from Placebo) BNC210 ng/ml

BNC210 Phase 2b PTSD ATTUNE Study Underway 19 WEEK 1 2 3 4 5 6 7 8 9 10 11 12 15 PHASE 2b OUTPATIENT BID DOSING FOLLOW-UP Single potential registrational- supporting trial for monotherapy Phase 2b treatment in PTSD 1:1 RANDOMIZED BNC 210 900 mg oral tablet DOUBLE-BLIND PLACEBO-CONTROLLED PLACEBO KEY INCLUSION CRITERIA BNC210 MONOTHERAPY IN PTSD PATIENTS Female and male (18 – 75 years) ~200 Subjects Current PTSD diagnosis CAPS-5 ≥ 30 (Screening & Baseline) SECONDARY ENDPOINTS PRIMARY ENDPOINT (& ≤ 25% decrease Screening to Baseline) Various patient-reported symptoms Investigator-rated PTSD symptoms of PTSD, changes in anxiety and on CAPS-5 Total Symptom Severity ~25 Sites depression symptoms, and global Scores in change from Baseline to and social functioning; Week 12 compared to placebo Safety & tolerability endpoints Topline data expected 1H’23 Fast Track designation from FDA BID = Twice daily dosing CAPS-5 = Clinician-Administered PTSD Scale for the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5)

20 CNS-focused Collaborations

Ongoing Strategic Collaboration with Merck & Co. 21 • Entered into in 2014 to develop α7 receptor PAMs targeting cognitive dysfunction associated with Alzheimer’s disease and other central nervous system conditions MSD • Merck funds all R&D activities including clinical development and WW commercialization of any products from collaboration Collaboration • Milestone payments of US$20M upfront and US$10M in 2017 when 1st compound entered Phase 1 clinical trials Overview • Eligible to receive up to US$465M in additional development and commercial milestone payments plus royalties • Includes 2 candidates which are PAMs of the α7 receptor in early-stage Phase 1 safety and biomarker clinical trials for treating cognitive impairment st Development • The 1 compound has completed Phase 1 safety clinical trials in healthy subjects and there are ongoing plans for further biomarker studies Updates • In 2020, a second molecule that showed an improved potency profile in preclinical animal models was advanced by Merck into Phase 1 clinical trials Snapshot of Early BNC375 Studies P A R T N E R S H I P Wang et al. J Pharmacol Exp Ther 373:311–324, May 2020 https://pubmed.ncbi.nlm.nih.gov/32094294/ PAM = Positive allosteric modulator MSD = A tradename of Merck & Co., Inc., Kenilworth NJ USA

22 MOU for BNC210 and MDMA Derivative (EMP-01) for Post-Traumatic Stress Disorder Joint Feasibility Assessment with: • Memorandum of Understanding (MoU) with EmpathBio Inc (EmpathBio) • To collectively explore a combination drug treatment regimen with BNC210 and EMP-01 22 February 2021 Illustrative • Initial collaborative framework of preclinical studies Memorandum of Understanding • MDMA-assisted psychotherapy has demonstrated with EmpathBio’s MDMA Derivative significant symptom improvement in PTSD patients • Initial collaborative framework of preclinical studies to collectively explore a • FDA has granted a Breakthrough Therapy designation to combinationdrugtreatmentregimenwithBNC210andEMP-01 MDMA-assisted psychotherapy. • MDMA-assisted psychotherapy has demonstrated significant symptom • EmpathBio is developing MDMA derivatives that may improvementinPTSDpatients permit the entactogenic effects of MDMA to be separated • FDA has granted a Breakthrough Therapy designation to MDMA-assisted psychotherapy EMP-01 = 3,4-Methylenedioxymethamphetamine from some of the known adverse effects. (MDMA) derivative • EmpathBio is developing MDMAderivatives that maypermit the entactogenic • To explore the possibility of a combination treatment effectsofMDMAtobeseparatedfromsomeoftheknownadverseeffects regimen warranting clinical evaluation • To explore the possibility of a combination treatment regimen warranting clinicalevaluation

23 Investment Highlights & Stock and Financial Information

1 Stock and Financial Snapshot 24 • Cash: US$40.4M / A$53.9M • Debt: $0 • Shares Outstanding: ~1,310M (NASDAQ:BNOX | ASX:BNO) • Warrants Outstanding: 142M (WAEP = US$0.04 / A$0.06) • Significant Investors: ▪ Biotechnology Value Fund ▪ Apeiron Investment Group Ltd. ▪ Merck & Co Figures as of September 30, 2021 unaudited financials, pro forma for Dec-21 US Offering and exercise of various options and warrants. Based upon the exchange rate of 1.33 as published by the Reserve Bank of Australia as of June 30, 2021. 1. Pro forma for ~16.1M out-of-the-money warrants expired on December 10, 2021.

Bionomics Outlook: Renewed Value-driving Trajectory 25 Diversified model with multiple value-driving clinical milestones expected in the next 4 – 6 quarters BNC210’s novel rapid onset formulation granted Fast Track designation for acute treatment of SAD 1 Established clinical proof-of-concept ; expect Phase 2 topline data by YE’22 BNC210 Phase 2b ATTUNE PTSD study under way with Fast Track designation for 1H’23 topline data Tablet formulation achieves exposure projected from pharmacometric analysis Merck strategic partnership for treatment of cognitive impairment in Alzheimer’s disease with two compounds in clinical development Diverse early-stage pipeline of partnering prospects targeting Kv and Nav ion channels for treatment of schizophrenia and pain, respectively Well-capitalized balance sheet driven by experienced leadership SAD = Social Anxiety Disorder PTSD = Post-Traumatic Stress Disorder 1. Wise T. et al., Biological Psychiatry 2020 (https://doi.org/10.1016/j.biopsych.2019.12.013); Perkins A. et al., Translational Psychiatry 2021 (https://doi.org/10.1038/s41398-020-01141-5)

26 APPENDIX: Management Team & Board of Directors

Powered by a Seasoned and Experienced Management Team 27 1 BOARD OF DIRECTORS Connor Bernstein Errol De Souza, PhD Errol De Souza PhD VP Strategy & Corporate Executive Chairman Executive Chairman Development David Wilson Non-Executive Director Adrian Hinton Alan Fisher Liz Doolin Non-Executive Director Interim Chief Financial VP Clinical Development Officer Jane Ryan PhD Non-Executive Director Aaron Weaver Apeiron Nominee Miles Davies Apeiron Nominee 1. Logos reflect experience in current and/or past roles.

28 APPENDIX: BNC210 Prior Clinical Trial Information

Summary of BNC210 Clinical Trials: Excellent Safety and Tolerability Profile in 29 Healthy Subjects and Patients Participants / Subjects Enrolled / Phase Description BNC210 Formulation and Doses Location Setting Administered BNC210* 1 Single Ascending Dose Safety and PK Healthy volunteers / In-clinic 32/24 Suspension; single doses (5 to 2000 mg) Australia 1 Single Ascending Dose Safety and PK; Food Effect Healthy volunteers / In-clinic 4/3 Suspension; single doses (300 to 2000 mg) Australia 47/40 1 Single Ascending Dose Safety and PK; Food Effect Healthy volunteers / In-clinic Capsule; single doses (300 to 3000 mg) US 1b Lorazepam Comparison Healthy volunteers / In-clinic 24/22 Suspension; single doses (300 and 2000 mg) France 1b CCK-4 Panic Attack Model Healthy volunteers / In-clinic 60/59 Suspension; single doses (2000 mg) France Multiple Ascending Dose Safety and PK; Expanded Suspension; multiple doses 1b Healthy volunteers / In-clinic 56/44 France Cohort for EEG Target Engagement (150 to 1000 mg twice daily for 8 days) 1 Suspension and Tablet Formulation PK Comparison Healthy volunteers / In-clinic 6/6 Suspension and tablet; single doses (300 mg) Australia 1 Single Ascending Dose Safety and PK Healthy volunteers / In-clinic 5/5 Tablet; single doses (600 to 1200 mg) Australia 1 Multiple Dosing Safety and PK Healthy volunteers / In-clinic 10/10 Tablet; multiple doses (900 mg twice daily for 7 days) Australia Imaging and Behavioral Study In Generalized Generalized anxiety disorder patients 2a 27/25 Suspension; single doses (300 and 2000 mg) UK Anxiety Disorder / In-clinic Suspension; multiple doses 2a Agitation in the Elderly in Hospital Setting Agitated elderly patients / Hospital 38/18 Australia (300 mg twice daily for 5 days) Post-traumatic stress disorder Suspension; multiple doses Australia 2 Post-Traumatic Stress Disorder 193/143 patients / Out-patient (150, 300 or 600 mg twice daily for 12 weeks) US Post-traumatic stress disorder Tablet; multiple doses 2b Post-Traumatic Stress Disorder Ongoing US patients / Out-patient (900 mg twice daily for 12 weeks) * The number of enrolled subjects who were administered BNC210; other enrolled subjects were administered placebo only CCK-4 = Cholecystokinin Tetrapeptide EEG = Electroencephalography PK = Pharmacokinetic

Mechanistic Validation of BNC210 as Evidenced by EEG Response 30 BNC210 Reduced Nicotine-induced EEG Responses Activation of nicotinic receptors in the brain induces EEG response BNC210 blood-brain α4β2 and α7 receptors are the major nAChR populations targeted barrier penetration and nicotinic receptor BNC210 daily oral dosing reduced nicotine-induced EEG in the α2 band target engagement in humans Observed reduction in EEG response due to BNC210’s negative allosteric modulation of the α7 receptors nAChR = Nicotinic Acetylcholine Receptor EEG = Electroencephalography * p-value less than 0.05 ** p-value less than 0.01

Phase 2 Trial of BNC210 in Adults with Post-Traumatic Stress Disorder (PTSD) 31 • Multi-center, randomized, double-blind, placebo-controlled • BNC210 150 mg, 300 mg, 600 mg and placebo (1:1:1:1) (liquid suspension formulation taken twice daily, b.i.d.) Study Design • 12-week treatment period • 193 participants • 20 US sites / 6 Australian sites • Current diagnosis of PTSD as defined by CAPS-5 (Clinician- Key Selection Criteria Administered PTSD Scale for DSM-5) • To assess the effects of BNC210 on investigator-rated symptoms of PTSD measured by CAPS-5 Key Study Objectives • To assess the safety and tolerability of BNC210 in subjects with PTSD

BNC210 PTSD Trial Overall Conclusions 32 No overall effect on primary endpoint of CAPS-5 total severity score at 12 weeks Australian patients had a greater improvement over placebo than US patients ✓CAPS-5 statistically significant at Week 4 in Australians (p<0.05) Evidence of antidepressant effect in high dose treatment group in total population ✓CAPS-5 Criterion D overall (negative alterations in cognitions and mood) statistically significant at Week 1 (p<0.05) ✓CAPS-5 Criterion D, Question 2 (persistent and exaggerated negative beliefs or expectations) statistically significant at Week 1 (p=0.001) ✓CAPS-5 Criterion D, Question 4 (persistent negative emotional state) statistically significant at Weeks 4 and 8 (p<0.05) Trend for anxiolytic effect in high dose treatment group in the total population ✓Trend towards improvement on CAPS-5 Criterion E (marked alterations in arousal and reactivity), Question 3 (hypervigilance) ✓Trend towards improvement on CAPS-5 Criterion E, Question 4 (exaggerated startle response) BNC210 was well tolerated in patients with PTSD ✓No trend for increased adverse events with treatment ✓No evidence of cognitive impairment ✓No evidence of suicidal ideation or behavior worsening Potential reasons why clinically significant effects and trends seen at early time points did not translate into significant primary endpoint on CAPS-5 at 12 Weeks • Inadequate overall blood exposure of BNC210 • Lower compliance with liquid suspension formulation which needed to be taken with food

33 Emerging CNS Pipeline for Partnering

Kv3.1 / Kv3.2 Ion Channel Activators for Cognitive Dysfunction and Negative Symptoms 34 Promising therapeutic strategy for Potential in schizophrenia, Autism Spectrum disorders improving cognitive disfunction and social and conditions with cognitive impairments withdrawal symptoms Bionomics’ molecules target Kv3.1/3.2 ion channels on parvalbumin positive, gabaergic interneurons in the pre-frontal cortex ~600 COMPOUNDS Lead Compound BL-76 Fully Reverses PCP-induced SYNTHESIZED Cognitive Deficit in Mice in the T-maze 8 0 100% -0% - -75% - 43% - 70% - 103% REVERSAL OF PCP EFFECTS 2 SERIES PATENTED 6 0 V e h i c l e P C P 3 m g / k g s c Lead Back-up 4 0 R i s p e r i d o n e 0 . 0 0 1 m g / k g i p / P C P Compound Compounds B L - 7 6 3 m g / k g p o / P C P BL-76 2 0 B L - 7 6 1 0 m g / k g p o / P C P 2 Patents Published B L - 7 6 3 0 m g / k g p o / P C P 0 S p o n t a n e o u s a l t e r n a t i o n ( % )

Pan Nav Inhibitors: Potential Non-Addictive, Reduced Side-Effect Chronic Pain Therapies 35 Associated with human pain Gain & Loss-of-function Disease-Related syndromes where extreme mutations in Nav1.7, 1.8 Genetics pain or no pain is experienced and 1.9 1000+ COMPOUNDS Lead Candidate SYNTHESIZED BNO Pan Nav inhibitors Identified Small molecules with 2 SERIES PATENTED BL-017881 functional selectivity for voltage gated sodium OBSERVED TO LEAD BACK-UP channels: Nav1.7, REVERSE PAIN IN COMPOUND COMPOUNDS BL-017881 THE FORMALIN PAW Nav1.8 and potentially MODEL IN MICE 3 Patents Published Nav1.9

36 APPENDIX: Building Value Through Legacy Oncology Assets

Legacy Oncology Asset Monetization to Drive Value: BNC101 37 Exclusive BNC101 Oncology License Agreement • Exclusive Agreement to license Bionomics’ BNC101 oncology for the Development of CAR-T Therapeutics drug candidate to Carina Biotech for the development of Chimeric Antigen Receptor T cell (CAR-T) therapy, which harnesses the body’s immune system to fight cancer. • Bionomics is eligible to receive up to A$118 million in clinical & development milestones plus royalty payments if Carina fully develops and markets the new therapy. In the event that Carina sub-licenses the CAR-T treatment, Bionomics is eligible to share in the sub-licensing revenues in early clinical development and receive a substantial double-digit portion of the revenues in later stages of clinical development. • In September 2021, Carina announced that it plans to initiate a clinical trial of BNC101 CAR-T therapy for the treatment of advanced colorectal (bowel) cancer in late 2022 • Bionomics retains BNC101 for other types of therapies
